Planet Earth/4d. Greenhouse Gases
How gasses interact with electromagnetic radiation
[edit | edit source]
All gases, including those in the atmosphere, reflect, scatter and absorb photons. Gasses are composed of molecules more widely spaced than molecules found in liquids and solids. When photons from sunlight pass through the atmosphere, these widely spaced gas molecules absorb some of the light, causing the atmosphere to block these sun’s rays, while some molecules let the higher energy light waves pass through the atmosphere, but block lower energy light waves that are typically reflected back into space. The molecules which absorb photons in the invisible lower energy infrared spectrum of light are collectively called Green House gasses, and include four key molecules found in Earth’s atmosphere: Water vapor (H2O), Carbon dioxide (CO2), Methane (CH4), and Nitrous oxide (N2O). There are other gas molecules which can absorb infrared light, including Ozone, Chlorofluorocarbons, and Hydrofluorocarbons, but these molecules do not absorb as much infrared light as the big four.
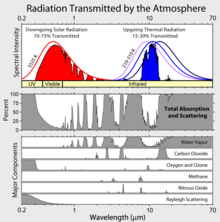
The Earth’s atmosphere is composed almost entirely of nitrogen (N2), oxygen (O2), and argon (Ar) which can block high energy light in the Ultraviolet or (UV) light spectrum, such as oxygen, but none of these gas molecules absorb infrared light. As such, none of them are considered Green House gasses.
Your eyes are perfectly adapted to seeing light in the visible spectrum within a narrow band of wavelengths (380 to 700 nanometers). The visible spectrum of light is the range of light waves from the sun that are able to pass through the atmosphere to reach the surface of the Earth. You cannot see Ultraviolet wavelengths (smaller than 380 nanometers) because so few UV light waves make it through the oxygen rich atmosphere. Likewise, water vapor (H2O) blocks much of the InfraRed wavelengths (greater than 1,000 nanometers) coming from the sun. Your eyes are adapted to a narrow slit of visible light wavelengths within the range permitted to pass through the Earth’s atmosphere. All the colors of the world; violet, indigo, blue, green, yellow, orange, and red, are of course not all the colors that exist, but are the range of wavelengths of light allowed to pass through the gasses that are found in Earth’s atmosphere. Water vapor (H2O) as a Green House gas is one of the most important. As a large molecule, water vapor in the air arises from evaporation and is measured by relative humidity.
Water Vapor as a Greenhouse Gas
[edit | edit source]
Water vapor (H2O) can absorb an enormous range of infrared wavelengths, with absorption bands around 1,000 nanometers, 6,000 nanometers and any infrared light longer than 12,500 nanometers. In fact, microwave ovens work because water (H2O) has such a broad band of absorption of light wavelengths that extend out to long microwave lengths, of several centimeters.
The amount of water vapor absorbed within the atmosphere is directly related to the temperature of the atmosphere. When the atmosphere is hot, the holding capacity of water vapor in the atmosphere increases, when the atmosphere is cold, the holding capacity of water vapor decreases. The amount of water vapor in air is directly related to temperature, but also pressure. Air with low pressures have less capacity to hold water vapor than higher pressures. This relationship will be explored further later in discussions of atmospheric pressure and weather patterns.

The relationship between temperature and the holding capacity of water can be observed on cold winter days. Imagine a cabin in the woods, which is heated by a gas stove inside the cabin. Outside air is cold, and hence holds less water vapor. However, when this outside air comes inside the cabin and is heated, the air suddenly increases its capacity to hold more water vapor. This undersaturated air will result in an increase in evaporation within the cabin. Water will be sucked into the warmed air, leaving any occupant inside the cabin with dry cracked skin. This also is the reason that some of the Earth’s coldest regions, are also some of Earth’s driest places. The cold air cannot hold as much water vapor as warm air.
This relationship is directly a result of water vapor being a Green House gas that absorbs infrared light waves. Inside the heated cabin, the amount of infrared light is much greater than outside the cabin. Because water absorbs this energy, the water vapor molecules will increase their energy states and undergo a phase transition from liquid water to a gas.
The term “Green House” is slightly a misnomer, as greenhouses trap heat inside by allowing sunlight to pass through transparent windows which prevent the exchange of air with the outside. Warm air inside the greenhouse can hold more water, and water tanks or sprinklers will make the inside of the greenhouse more humid for plants to grow. If a greenhouse does not contain any water source, it becomes very dry inside during the winter, and would likely dry out any plants left in such an environment. Greenhouses work best with the availability of the Green House gas of water vapor, which is able to absorb the infrared light, making greenhouses particularly humid and warm, and ideal for growing plants.
Methane as a Greenhouse Gas
[edit | edit source]
Methane (CH4) is another Green House gas, which absorbs infrared light, with a specific range around 3,000 nanometers and between 7,000 to 8,000 nanometers in the infrared spectrum of light. Methane is a particular strong Green House gas, because of the band of absorption between 7,000 to 8,000 which is not absorbed by water vapor. An atmosphere rich in both water vapor and methane can absorb a wider range of infrared wavelengths of light, allowing less light to escape to space, and retaining more of that energy near the surface of the Earth. Methane is typically a rare gas in the atmosphere, but recent measurements of methane have shown a dramatic increase from 1000 ppb in the 1950s to 1900 ppb in 2020. Ice cores indicate that methane was never above 800 ppb until the modern age. Indicating that since the advent of humans (Homo sapiens) on Earth, methane in the atmosphere has more than doubled. Since 2007, methane has increased dramatically, particularly in the northern hemisphere, as observed from the atmospheric infrared sounder (AIRS) aboard NASA’s Aqua satellite.
Methane under the Arctic region forms vast deposits of methane hydrate, a solid state of methane that forms under pressure and cold temperatures. In the deep cold ocean waters of the Arctic Ocean, methane accumulates due to the decay and breakdown of ocean organisms, and methanogens which feed on this organic carbon and release methane. The intense cold ocean water (near freezing temperatures), and intense pressure of the deep water, results in a solid form of methane to accumulate on the ocean floor, called methane hydrate (also called methane clathrate). If ocean waters warm, this methane outgases with a phase transition from a solid to a gas, and this gas bubbles up from the ocean floor and into the atmosphere.
Methane in the presence of oxygen results in the reaction CH4 + 2O2 → 2H2O + CO2 an oxidization of methane that occurs in the atmosphere. Note, that this reaction results in the production of both water vapor and carbon dioxide, two other greenhouse gasses. The resistance time of methane in the atmosphere is short about 8 to 9.5 years. This indicates that today’s high values of methane in the atmosphere are less a culminative effect, but represent net yearly increases of methane into the atmosphere. The atmospheric infrared sounder (AIRS) aboard NASA’s Aqua satellite gives a grim picture of the North Hemisphere of Earth, with a recent enrichment of the atmosphere of methane. The source of the methane has alarmed atmospheric scientists.
In addition to methane outgassing from the High Arctic, methane outgasses from oil and gas fields which frequently leak methane from wells and when transported in gas pipes. Technology advancements of fracking rock to release underground naturally occurring methane deposits, which are used as a natural gas fuel source and coal-bed methane; the process of using chemicals (acids) on underground coal to release methane fuel have likely led to some of the recent increases in methane in the atmosphere. Livestock, such as dairy farms also contributes to methane in the atmosphere. Using infrared sensors (both from aircraft and on the ground) scientists have found some of these sources, and governments have passed laws to curb the industrial release of methane into the air that we breathe. Methane in the atmosphere remains a major concern for atmospheric scientists.
Carbon dioxide as a Greenhouse Gas
[edit | edit source]
Carbon dioxide has become synonymous with greenhouse gas. As a large molecule carbon dioxide absorbs infrared light around 4,250 nanometers and a broad band between 13,900 to 16,100 nanometers. This longer wavelength band overlaps water vapor, but in cold dry air, high carbon dioxide can have a dramatic effect on the absorption of infrared light. The lower band around 4,250 nanometers is within an infrared band that most other greenhouse gasses do not absorb, making carbon dioxide, in the addition to methane and water vapor a very effective greenhouse gas. Since 1958 Carbon dioxide has risen from 310 ppm to 410 ppm in 2020, and with a very long resistance time in the atmosphere (several hundred thousand years), carbon dioxide released into the atmosphere has been accumulating at an alarmingly fast rate. Naturally carbon dioxide can be drawn down from the atmosphere by reacting with H2O to form carbonic acid, which becomes bonded to calcium bearing rocks, as calcium carbonate, but this process is very slow, at rates that mountains slowly erode. A faster method of removal of carbon dioxide from the atmosphere results from photosynthesizing life forms, which need to be buried underground after death, otherwise the carbon dioxide can be re-released into the atmosphere if these lifeforms decay or are combusted in fires, such as forest fires, which can release massive amounts of carbon dioxide back into the atmosphere.
Nitrous oxide as a Greenhouse Gas
[edit | edit source]
Nitrous oxide (N2O) is an important greenhouse gas that does not get as much attention as methane and carbon dioxide. Nitrous oxide gas spans a number of absorption bands in the infrared spectrum, and hence is a greenhouse gas. Nitrous oxide levels have been also increasing in the atmosphere from 288 ppb to about 330 ppb in 2020. Much of this nitrous oxide comes from a complex process of the oxidation of ammonia (NH3) used as fertilizers on crops, as well as from combustion engines, as nitrous oxide allows the engines to burn more fuel by providing more oxygen than air alone, resulting in a more powerful combustion. Urban centers, particularly where automobile traffic is high, often have higher concentrations of nitrous oxide.

The process of oxidation of ammonia (NH3) is complex, and involves NH3-oxidising bacteria, which take ammonia (NH3) and oxidise the nitrogen to nitrite (NO−2), followed by the reduction to nitric oxide (NO), which forms N2O, and finally nitrogen (N2). Plants require nitrogen to grow, but are unable to use atmospheric molecular nitrogen (N2). Nitrogen fixation by bacteria in the soils, help produce ammonia (NH3) that plants require. The industrialization of agriculture has pushed for the widespread application of human produced ammonia (NH3) on crops to increase plant growth. This has led to increases in nitrogen oxide compounds, often called NOx (often referred to as nox, or nitrogen oxides). Unlike carbon dioxide, the nitrogen oxides absorb some light waves in the visible spectrum, and produced brown clouds of smog, when concentrations are high above urban centers. Nitrogen oxides are also related to the formation of near surface ozone, which is a toxic gas. This process requires visible light from the sun (wavelengths around 420 nanometers), via the Leighton relationship. Ozone levels at the surface of Earth are closely monitored because when they are elevated they cause damage to the tissues of the lungs.
All these greenhouse gasses absorb some amount of the infrared spectrum of light, resulting in capturing energy that in their absence would have been reflected back into space. Greenhouse gasses act like a wool blanket, preventing Earth’s heat from escaping into space. The more Greenhouse gasses are in the atmosphere, the greater the thermal insulation of the planet. Higher global temperatures result in great amounts of water vapor in the atmosphere, a powerful greenhouse gas; a positive feedback that results in even higher global atmospheric temperatures. Methane is released from the deep cold oceans when temperatures in the deep ocean increase, resulting in another powerful feedback that further results in even higher global atmospheric temperatures. A vicious cycle can quickly develop that would lead to runaway global temperatures, and such runaway greenhouse outgassing events have happened in Earth’s past 55.5 million years ago, at the Eocene-Paleocene Boundary (the PETM event), and 252 million years ago, at the Permian-Triassic Boundary (during the Great Dying event). Such events significantly altered the Earth, making it less habitable for animal life.
| Previous | Current | Next |
|---|---|---|
