Radiation Oncology/Retinoblastoma
Appearance
|
Epidemiology
[edit | edit source]
- Most common primary ocular malignancy of childhood; ~4% of pediatric malignancies
- Incidence 1/20,000
- ~250 cases per year in USA
- Higher rates in developing countries
- Bilateral disease 20-30%
- 10% patients have family history; 90% are sporadic (of these 20-30% are bilateral, and therefore heritable)
- Overall 40% have hereditary retinoblastoma, 60% have sporadic type
- Average age at diagnosis 1 year for bilateral; 2 years for unilateral
- Uncommonly, patients with bilateral retinoblastoma can also develop a midline neuroblastic tumor. The syndrome is called "trilateral retinoblastoma" (75% of the third tumors are pineoblastoma)
- Heritable form transmitted in autosomal dominant fashion
- Clinical presentation
- Leukocoria (white reflection in the pupil): initially inconstant, visible at certain angles/light conditions, may be seen on flash photography
- Strabismus
- Also iris rubeosis, hypopyon, hyphema, orbital cellulitis, and exophthalmia
- Some children may have no symptoms
Diagnosis
[edit | edit source]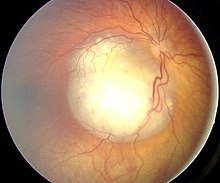

- Awake examination with attention to visual acuity, extraocular movement, pupillary examination, slit-lamp examination, and indirect ophthalmoscopy
- Ocular fundus examination under general anesthesia: white tumor with angiomatous dilation of the vessels
- Unilateral or bilateral nature of the lesions
- Number of tumors
- Location in the retina (posterior pole, anterior retina), and anatomica relations with optic disc and macula
- Tumor size (diameter, thickness)
- Subretinal fluid and tumor seeds
- Vitreous seeding (localized vs diffuse)
- Ocular ultrasound: On A-mode shows very intense reflections from calcium deposits. On B-mode shows a heterogenous acoustic solidity with highly reflective intrinsic echoes within the tumor and attenuation of orbital pattern
- Differential diagnosis includes Coats' disease, retinal detachment, retinopathy of prematurity, persistent hyperplastic primary vitreous
- MRI: assessment of local extension to optic nerve, anterior chamber, and orbital fat. Slightly hyperintense on T1, hypointense on T2. Also to rule out trilateral retinoblastoma
- CT: intraocular mass with a higher density than the vitreous body, calcified in 90% of cases and moderately enhanced after iodine contrast agent injection. However, should be minimized due to ionizing exposure, particularly in heritable cases
- If patients needs enucleation, consider systemic staging with CSF cytology, bone marrow cytology, and spinal axis MRI
- Genetic testing should be considered
Pathology
[edit | edit source]
- First disease for which genetic etiology was demonstrated (Knudson, 1971 - two hit hypothesis)
- Rb gene located on 13q14 is part of G1/S checkpoint. It is also required for appropriate exit from the cell cycle of retinal progenitor cells and for rod development
- >900 mutations of Rb gene have been reported, many in areas of binding domain with E2F
- However, other molecular events are also necessary for tumorigenesis; n-myc is amplified in ~10%
- Given higher prevalence in developing countries, HPV (Human Papilloma Virus, via E7) may play a role
- Neuroepithelial origin
- Tumor cells form Flexner-Wintersteiner rosettes
Staging
[edit | edit source]- Historically, retinoblastoma was separated into intraocular and extraocular disease
- Intraocular retinoblastoma was classified using the Reese-Ellsworth staging, which was based on the likelihood of preserving the eye after EBRT. However, it does not have good predictive power in strategies involving initial chemotherapy
- International Classification System was developed in 2003 to predict outcomes in response to chemotherapy and focal consolidative therapy, and is used in COG protocols
Reese Ellsworth Staging
- Stage Ia - solitary, <4 disc diameters, at or behind equator
- Stage Ib - multiple, <4 disc diameters, at or behind equator
- Stage IIa - solitary, 4-10 disc diameters, at or behind equator
- Stage IIb - multiple, 4-10 disc diameters, at or behind equator
- Stage IIIa - anterior to equator
- Stage IIIb - solitary tumor >10 disc diameters, behind equator
- Stage IVa - multiple tumors, some >10 disc diameters
- Stage IVb - anterior to ora serrata (serrated jct. between retina & ciliary body)
- Stage Va - massive tumor, > 50% retina
- Stage Vb - vitreos seeding
International Classification System
| Group | Characteristics |
|---|---|
| Group A (Small) |
<= 3mm height, >= 3mm from fovea, >= 1.5 mm from optic nerve |
| Group B (Medium) |
>3mm height, clear subretinal fluid <= 3mm from tumor margin |
| Group C (Confined Medium) |
Localized vitreous seeding (C1), subretinal seeding <= 3mm from tumor margin (C2), or both (C3) |
| Group D (Diffuse Large) |
Diffuse vitreous (D1) or subretinal seeding >3mm from tumor margin (D2), or both (D3). Subretinal fluid >3mm from tumor margin |
| Group E (Advanced) |
No visual potential, or presence of >=1 of the following: tumor in anterior segment, tumor in ciliary body, neovascular glaucoma, vitreous hemorrhage, phthisical eye, orbital cellulitis-like presentation, involvement of optic nerve, extraocular disease on neuroimaging |
Treatment
[edit | edit source]- For small localized tumors, enucleation or EBRT were used historically. EBRT use declined recently due to significant late toxicity and risk of second tumors. There is now increasing use chemotherapy and focal therapies such as thermotherapy, photocoagulation, cryotherapy, and plaque brachytherapy
- For extensive unilateral lesions, enucleation or definitive radiation are the main approaches
- Indications for adjuvant treatment after enucleation are somewhat controversial, but may include optic nerve involvement posterior to the lamina cribrosa as an independent finding; posterior uveal invasion (includes choroidal invasion); any degree of concomitant choroid and optic nerve involvement; tumor involving the optic nerve posterior to the lamina cribrosa (passage through sclera) as an independent finding; scleral invasion; anterior chamber seeding; ciliary body infiltration; iris infiltration
- COG has an ongoing study of adjuvant carboplatin, etoposide, and vincristine x 6 cycles in patients considered at "high risk" after enucleation (ARET0332) for the following high-risk criteria:
- Massive choroid replacement defined as posterior uveal invasion grades IIC and IID
- Any posterior uveal involvement with any optic nerve involvement (optic nerve head, prelamina and post lamina cribrosa (Note: both posterior uveal involvement AND optic nerve involvement are required)
- For bilateral disease, conservative approaches have been suggested, but depend on size, number, and location of lesions
- Chemoreduction, with enucleation of the "worse" eye and focal therapy of the "better" eye can be considered
- Patients with extraocular retinoblastoma have a poor prognosis, although high-dose chemo with stem cell rescue is being explored by COG
- Treatment Guideline based on UCSF Protocol (PMID 19477707, 2009)
- Group A: Focal therapy only (laser, cryotherapy, hyperthermia, brachytherapy)
- Group B: Vincristine + carboplatin up to 6 cycles; focal therapy with 2-6 cycles
- Group C: Vincristine + carboplatin + etoposide up to 6 cycles; focal therapy
- Group D: Same as C. EBRT
- Group E: Enucleation. Prophylactic 3-agent chemotherapy
External Beam Radiation Therapy
[edit | edit source]- Historically used as a treatment alternative to avoid enucleation in select patients
- Target was entire retina, up to ora serrata anteriorly
- Initially, "D-shaped" field was used
- Alternatively, a wedge pair with anterior and lateral fields could be used
- To obtain better sparing of lens, anterior lens block could be added
- Local control rates were >75% for early lesions
- However, long-term toxicity was significant. In particular, stunted growth of orbital bone and poor long term cosmetic outcome became a problem
- Orbital bone growth centers are located along the sutures, frontozygomatic in lateral wall, frontomaxillary in the medial wall, and zygomatic-maxillary in the orbital floor
- Also, for patients with hereditary retinoblastoma, risk of second malignancy has been reported as high as 51% at 50 years
- Efforts are now under way to avoid EBRT in the primary treatment setting, and use it for salvage only
- Two dosimetric studies have shown that proton therapy may be dosimetrically superior to photon EBRT in settings where focal therapies are not appropriate
- MSKCC, 1996 (1979-84) - PMID 8641925 -- "External beam radiation therapy and retinoblastoma: long-term results in the comparison of two techniques." Int J Radiat Oncol Biol Phys. 1996; 35(1):45-51
- 123 pts, 1979-91, tx'd w/ primary EBRT (larger fx size, 2.5 Gy, used b/f 1984), range of xrt 38-50 Gy
- Series compared a modified lateral beam technique to an anterior lens sparing technique; local control for Group I-III tumors appeared to be improved w/ modified lateral beam (84% vs 38% at 5 yrs)
- Hahnemann, 1996 (1980-1991) - PMID 8641908 -- "External beam radiation for retinoblastoma: results, patterns of failure, and a proposal for treatment guidelines." Int J Radiat Oncol, Biol, Phys. 1996; 35(1):125-32
- 27 pts, 34 eyes, RE Groups I-V tx'd w/ definitive EBRT (median 45 Gy)
- Local control 78.5% in RE Groups I-II, 20% in RE III-V; EBRT did NOT appear to prevent new tumors in clinically uninvolved retina.
- St. Jude's, 1969 PMID 5822720 Cassady, Ellsworth et al. "Radiation therapy in retinoblastoma. An analysis of 230 cases." Radiology. 1969; 93(2):405-9
- 230 pts, largest US series of retinoblastoma pts treated w/ EBRT. 3.3-4.0 Gy delivered 3x per week.
- Local control 73% in RE Groups I-II, 20% in RE III-V.
- No significant difference in local tumor control when doses of 32.5-35 Gy compared to 40-45 Gy.
Plaque Brachytherapy
[edit | edit source]- Plaque brachytherapy introduced in 1931 with radon seeds
- I-125 used commonly in the U.S., Ru-106 in Europe
- For Ru-106 plaques, tumors should be <5 mm deep due to steeper dose fall-off
Iodine-125
- Thomas Jefferson
- Salvage; 2006 (1994-2005) PMID 16949158 -- "Iodine 125 plaque radiotherapy as salvage treatment for retinoblastoma recurrence after chemoreduction in 84 tumors." (Shields CL, Ophthalmology. 2006 Nov;113(11):2087-92. Epub 2006 Sep 1.)
- Retrospective. 84 tumors in 71 eyes in 64 patients, solid recurrences after chemoreduction (vincristine, etoposide, carboplatin x6 cycles + thermotherapy or cryotherapy). I-125 plaque, dose 40 Gy to tumor surface + 2 mm margin. Mean thickness 4 mm. Mean foveola dose 45 Gy, optic disc 22 Gy. Prior EBRT in 30%. Median F/U 4 years
- Outcome: Local control after chemoreduction 95%, after chemoreduction + EBRT 100%
- Toxicity: Proliferative retinopathy 17%, maculopathy 24%, cataract chemoreduction 19% vs chemoreduction + EBRT 42%, iris neovascularization 8%
- Conclusion: I-125 plaque BT offers excellent tumor control; complications should be anticipated
- 2001 (1976-1999) PMID 11713089 -- "Plaque radiotherapy for retinoblastoma: long-term tumor control and treatment complications in 208 tumors." Ophthalmology. 2001; 108(11):2116-21.
- Retrospective. 141 children managed w/ plaque brachy; 71% received prior therapy (chemo, EBRT, cryotherapy, photocoagulation, etc); 35% s/p prior tx failure
- Outcome: 5-year local control 79%, if primary therapy 88%
- Toxicity: 27% non-proliferative retinopathy, 15% proliferative retinopathy, 31% cataracts, 0% scleral necrosis at 5 yrs
- Conclusion: Good tumor control, particularly for those that fail prior therapy
- Salvage; 2006 (1994-2005) PMID 16949158 -- "Iodine 125 plaque radiotherapy as salvage treatment for retinoblastoma recurrence after chemoreduction in 84 tumors." (Shields CL, Ophthalmology. 2006 Nov;113(11):2087-92. Epub 2006 Sep 1.)
- University of Cape Town; 2002 PMID 12459368. "Postenucleation orbits in retinoblastoma: treatment with 125I brachytherapy." Int J Radiat Oncol Biol Phys 2002; 54(5):1446-54
- 57 cases b/w 1983-2000, treated w/ post enucleation brachy; 34 Gy (w/ 30 cases also receiving chemo); 21 cases w/ +margins, extrascleral tumor or mets
Ruthenium-106
- Lausanne, Switzerland; 2008 (1992-2006) PMID 18207660 -- "(106)Ruthenium brachytherapy for retinoblastoma." (Abouzeid H, Int J Radiat Oncol Biol Phys. 2008 Jul 1;71(3):821-8. Epub 2008 Jan 22.)
- Retrospective. 39 children, 41 eyes, 63 tumors. First line treatment 5%, second-line 21%, salvage 74%. Plaque diameter >= 2 mm than tumor diameter, margin 1 mm to tumor height for sclera. Prescription dose target 50 Gy at apex. Minimum F/U 1 year
- Outcome: 1-year tumor control 73%. Recurrence 12% if 1st/2nd line, and 32% if salvage. Eye retention 76% (though 3/10 removed eyes for different tumor than the one treated)
- Toxicity: Retinal detachments 17%, proliferative retinopathy 2%, cataract 10%
- Conclusion: Ru-106 brachytherapy effective
Proton Therapy
[edit | edit source]- Paul Scherrer Institut; 2008 PMID 18290958 -- "New developments in external beam radiotherapy for retinoblastoma: from lens to normal tissue-sparing techniques." (Munier FL, Clin Experiment Ophthalmol. 2008 Jan-Feb;36(1):78-89.)
- Case report of 6 patients, and literature review
- MD Anderson; 2005 PMID 16168831 -- "Treatment planning with protons for pediatric retinoblastoma, medulloblastoma, and pelvic sarcoma: how do protons compare with other conformal techniques?" (Lee CT, Int J Radiat Oncol Biol Phys. 2005 Oct 1;63(2):362-72.)
- Treatment planning. 8 patients (3 Rb, 2 MB, 3 pelvic sarcoma), retrospective DVH comparison of 3D-CRT, electrons, IMRT, protons
- Retinoblastoma: Protons best coverage combined with most orbital bone sparing (>5 Gy protons 10%, electrons 25%, 3D lateral beam 41%, 3D anterolateral beam with lens block 51%, 3D anterolateral beam without lens block 65%, IMRT 69%). Single appositional electron field next best technique. 3D-CRT techniques significantly inferior
- Medulloblastoma: Protons least dose to cochlea, hypothalamus/pituitary. IMRT second best for posterior fossa. For craniospinal, 3D electrons better than 3D photons
- Pelvic sarcoma: Protons superior for ovary dose
- Conclusion: Protons superior, both for target coverage and for normal structure sparing
- Harvard
- 2006 Abstract Link -- "The Use of Gantry Decreases Normal Tissue Exposure in Proton Beam Radiotherapy of Intraocular Retinoblastoma" (Ciralsky JB, Invest Ophthalmol Vis Sci 2006;47: E-Abstract 2815.)
- Case report. Historically treated 35 patients using lateral beam. First 2 patients treated with gantry using anterolateral oblique beam.
- 2005 PMID 15667981 -- "Proton radiation therapy for retinoblastoma: comparison of various intraocular tumor locations and beam arrangements." (Krengli M, Int J Radiat Oncol Biol Phys. 2005 Feb 1;61(2):583-93.)
- Treatment planning. Different retinoblastoma tumor locations (posterior-central, nasal, temporal), different eye positions (straight, intrarotated, extrarotated), different beam arrangements (lateral, anterolateral oblique, anteromedial oblique)
- Conclusion: Homogenous target coverage, true lens sparing. Doses to orbit structures minimized depending on tumor location
- 2006 Abstract Link -- "The Use of Gantry Decreases Normal Tissue Exposure in Proton Beam Radiotherapy of Intraocular Retinoblastoma" (Ciralsky JB, Invest Ophthalmol Vis Sci 2006;47: E-Abstract 2815.)
Secondary Malignancies after RT
[edit | edit source]- New York Hospital PMID 9544627 "Second nonocular tumors in survivors of bilateral retinoblastoma: a possible age effect on radiation-related risk." Ophthalmology. 1998;105(4):573-9
- Retrospective review of 816 pts w/ bilateral retinoblastoma in NY area.
- 2nd malignancies more common if pts tx'd w/ EBRT before age 12 mo's.
- NCI Analysis; 1997 (1914-1984) PMID 9333268 "Cancer incidence after retinoblastoma. Radiation dose and sarcoma risk." (Wong FL, JAMA. 1997 Oct 15;278(15):1262-7.)
- Cohort study, NY and MA hospitals. 1604 cases of retinoblastoma, survived >1 year. 961 hereditary
- 2nd cancer incidence at 50 years: if germline mutation 51% (RR 30), if sporadic 5% (not elevated)
- Sarcoma risk: related to dose; threshold 5 Gy, 11x increase at 60+ Gy
- Conclusion: Genetic predisposition substantial impact on risk of subsequent cancers
Trilateral Retinoblastoma
[edit | edit source]- Sao Paulo; 2007 (Brazil)(1986-2003) PMID 16572402 -- "Trilateral retinoblastoma." (Antoneli CB, Pediatr Blood Cancer. 2007 Mar;48(3):306-10.)
- Retrospective. 470 children with Rb, 4 with pineoblastoma (2/4 with family history). 3 had bilateral disease
- Outcome: All died within 12 months
- Conclusion: New therapeutic approaches needed
Review
[edit | edit source]- UCSF; 2009 PMID 19477707 -- "Frontiers in the Management of Retinoblastoma." (Lin P, Am J Ophthalmol. 2009 May 23.)
