Structural Biochemistry/Apoptosis
Overview
[edit | edit source]Apotosis is a normal process during development and removing cells. These cells can develop to be cancerous cells.
Apoptosis is the programmed death of a cell. It occurs naturally in multicellular organisms and is used to regulate dead cells. Apoptosis occurs daily in human beings destroying naturally anywhere between 20 to 70 billion cells and those cells end up being replaced by newer cells.
During Apoptosis, cells will:
- Shrink
- Develop bubble-like blebs on their surface
- Have the chromatin in their nucleus degraded
- Have their mitochondria break down with the release of cytochrome c
- Break into small, membrane-wrapped, fragments
- Release ATP and UTP
- Their nucleotides will bind to receptors on phagocytic cells and this will serve as a signal to attract dying cells
- The phospholipid phosphatidylserine will be exposed on the surface
- The exposed phospholipid will serve as a “eat me” signal, which will induce the phagocytes to engulf the cell fragments
- The phagocytic cells secret cytokines that inhibit inflammation
This orderly pattern of events is called programmed cell death, also known as apoptosis.[1]
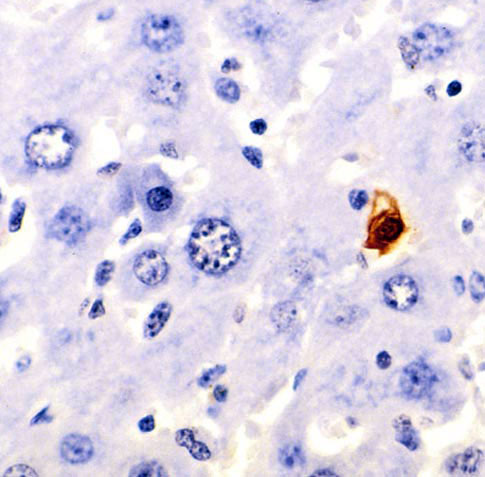
Apoptosis was discovered by German scientist Carl Vogt who first detailed the events of apoptosis. This discovery was then later given a more precise definition by Walther Flemming. Since the 1990s apoptosis has started to become more in the mainstream and being investigated more. It truly started in the 1960s when it was seen occurring under the microscope. The picture below is mouse liver cells and the one stained is going through apoptosis.
Since the pathway through apoptosis is very long if one biochemical reaction is not carried out it can eventually lead to cells that do not die and go on creating havoc in the organism. If the cells do not go through this natural phase in their life cycle they can end up causing diseases and/or disorders. This problem can be linked to the creation of cancer by a malfunctioning cell being able to reproduce and pass on any bad organelles or incorrect DNA sequences.
Below is a picture from the Wikimedia Commons briefly showing a depicted drawing of how the apoptosis occurs in the cell:
Apoptosis was studied in roundworms and in the final stages of its cell death, there discovery of the death-inducing protease CED-3 activation became the cause of the apoptosis. In order to regulate these proteases, CSP-3 was discovered to block CED-3 autoactivation and ultimately, to decrease signs of apoptosis during the developmental stages of roundworms. CSP-3 is a capspase homolog which only works in somatic cells. Therefore, germ cells are not affected by the CSP-3.
Necessity of apoptosis in cells
[edit | edit source]- Apoptosis is needed for the proper development and formation of body parts. For example, the formation of fingers and toes of a fetus is made possible by apoptosis of the tissue between them.
- Apoptosis is needed to destroy cells that pose threats to the well-being of the organism as a whole. For example, the destruction of cell with damaged DNA.
Required factors for cell's apoptosis to occur
[edit | edit source]- There is a withdrawal of positive signals. Cells need to receive continuous stimulation, positive signals, from other cells or they need a continuous adhesion to a surface in order to survive.
For example: The withdrawal of growth factors of neurons will cause apoptosis because the growth factors serve as positive signals and are crucial for cell development
- There is a receipt of negative signals.
For example: The increase of oxidants inside the cell or the accumulation of misfolded proteins.
The mechanism of apoptosis
[edit | edit source]There are 3 different mechanisms for cell apoptosis.
Apoptosis triggered by internal signals: the intrinsic or mitochondrial pathway
[edit | edit source]- In a healthy cell, the protein Bcl-2 is displayed on the surface of the mitochondria’s outer membrane. This protein inhibits apoptosis.
- Internal damage to the cell causes the protein, Bax, to migrate to the surface of the mitochondria and inhibits the effect of Bcl-2 and stick to the outer membrane surface of the mitochondria. Bax punches holes in the outer membrane and causes cytochrome c to spill out.
- Cytochrome c uses the energy provided by ATP and proceeds to bind to the protein Apaf-1 (“apoptotic protease activating factor-1”).
- The binding of cytochrome c to Apaf-1 forms apoptosomes.
- Apoptosomes bind to caspase-9 and activates it. Caspase-9 is a protease that cleaves proteins.
- As caspase-9 cleaves proteins, it activates other caspases from its family. Such as caspase-3 and caspase-7.
- A cascade of proteolytic activity is created by the activation of caspases. These proteolytic activities lead to the digestion of structural proteins in the cytoplasm, the degradation of chromosomal DNA, and the phagocytosis of the cell.[2]
Apoptosis triggered by external signals: the extrinsic or death receptor pathway
[edit | edit source]- Integral membrane proteins, such as Fas and TNF receptor, have receptors that are exposed at the surface of the cell.
- Binding of the complementary death activator, such as FasL and TNF, sends a signal to the cytoplasm that leads to the activation of caspase-8.
- Caspase-8, similar to caspase-9, activates a cascade of proteolytic activities, which leads to the phagocytosis of cell.[3]
Apoptosis-Inducing Factor (AIF)
[edit | edit source]- Neurons undergo apoptosis without the use of caspases.
- AIF is a protein that is located in the intermembrane space of the mitochondria.
- When a cell receives its death signal, AIF, which is released from the mitochondria, migrates into the nucleus and binds to the DNA, which then triggers the destruction of DNA.
- Beside programing cell death, AIF also plays a important mitochondrial role in healthy cells. "A segment of AIF which is dispensable for its apoptotic function carries an NADH-oxidase domain that regulates the respiratory chain complex I and is required for cell survival, proliferation and mitochondrial integrity."[4]
Researched results in Mitochondrial functions of AIF
[edit | edit source]Recent researches using mutant mice have helped in understanding of mitochondrial functions of AIF. For example, AIF-deficent mice are used as a model of complex I deficiency which shows a general reprogramming of mitochondrial metabolism.[5] In human, the deficiency of complex I causes >30% of mitochondrial diseases,[6] and it mainly affects on infants which shows a variety of symptoms such as epileptic seizures, blindness, deafness, axaxia, cognitive deficiency, myopathy and cardiomyopathy.
Researches have shown that the uncharacterized splice isoforms of AIF whose tisue and cell type-specific expression pattern can be the cause of some tissue-specific effects in AIF deficiency. In addition, the failure in detecting AIF mutations might lead to the embryonic lethality of AIF deficiency, which observed in mutant mice.[7]
Research have also shown than a 80% reduction in the AIF expression allows the development of Hq in mice and affects on health of adult animals due to an aggravating complex I deficiency.[8]
Role of AIF in the survival, proliferation and metabolism of cells
[edit | edit source]Mutant mice are used in studies of role of AIF in survival, proliferation and metabolism of cells. If level of AIF presence is low, it will not affect the inheritance of Hq mutation and has no major effect in exhibit growth retardation. An reduction of AIF level over time reduces major effects on the health of aging adult mice.[9] The complete loss of AIF would cause the incompatibility with intrauterine development, and also tend to cause AIF-null mice due to the failure of homologous recombination.[10]
Applications
[edit | edit source]Apoptosis and Organ Transplants
[edit | edit source]- Certain body parts, such as the testes and the anterior chamber of the eye, are not susceptible to antigen invasions. It turns out that these cells produce FasL at high rates, therefore, antigens, which produce Fas, would be killed as soon as they enter these sites.
- This discovery will potentially aid transplant recipients greatly, because the transplanted organs will less likely be susceptible to the attacks of the host body’s cell-mediated immune system if artificial cells made to produce high levels of FasL can be effectively inserted into the transplanted organs.
- So far, clinical trials on animals have been conducted. Allografts synthesized to express FasL have shown increases survival rate for kidneys but not for hearts.
Apoptosis in Plants
[edit | edit source]- Plants too undergo programmed cell death. Apoptosis may aid the plant in halting the spread of a virus infection.
- The mechanism of apoptosis in plants also involves caspases, which cleaves at the Asp residue. However, the activation of caspases destroys the central vacuole, which disintegrates the rest of the cell, and not the DNA.
Reference
[edit | edit source]- ↑ http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/A/Apoptosis.html
- ↑ http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/C/caspase9.png
- ↑ http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/C/CTL_Fas.gif
- ↑ Hangen, Emilie; Blomgren, Klas; Bénit, Paule; Kroemer, Guido; Modjtahedi, Nazanine (2010). "Life with or without AIF". Trends in Biochemical Sciences. 35 (5): 278–87. doi:10.1016/j.tibs.2009.12.008. PMID 20138767.
- ↑ Pospisilik, J. Andrew; Knauf, Claude; Joza, Nicholas; Benit, Paule; Orthofer, Michael; Cani, Patrice D.; Ebersberger, Ingo; Nakashima, Tomoki; Sarao, Renu; Neely, Gregory; Esterbauer, Harald; Kozlov, Andrey; Kahn, C. Ronald; Kroemer, Guido; Rustin, Pierre; Burcelin, Remy; Penninger, Josef M. (2007). "Targeted Deletion of AIF Decreases Mitochondrial Oxidative Phosphorylation and Protects from Obesity and Diabetes". Cell. 131 (3): 476–91. doi:10.1016/j.cell.2007.08.047. PMID 17981116.
- ↑ Koene, S.; Smeitink, J. (2009). "Mitochondrial medicine: Entering the era of treatment". Journal of Internal Medicine. 265 (2): 193–209. doi:10.1111/j.1365-2796.2008.02058.x. PMID 19192036.
- ↑ Vahsen, Nicola; Candé, Céline; Brière, Jean-Jacques; Bénit, Paule; Joza, Nicholas; Larochette, Nathanael; Mastroberardino, Pier Giorgio; Pequignot, Marie O; Casares, Noelia; Lazar, Vladimir; Feraud, Olivier; Debili, Najet; Wissing, Silke; Engelhardt, Silvia; Madeo, Frank; Piacentini, Mauro; Penninger, Josef M; Schägger, Hermann; Rustin, Pierre; Kroemer, Guido (2004). "AIF deficiency compromises oxidative phosphorylation". The EMBO Journal. 23 (23): 4679–89. doi:10.1038/sj.emboj.7600461. PMC 533047. PMID 15526035.
- ↑ Hangen, Emilie; Blomgren, Klas; Bénit, Paule; Kroemer, Guido; Modjtahedi, Nazanine (2010). "Life with or without AIF". Trends in Biochemical Sciences. 35 (5): 278–87. doi:10.1016/j.tibs.2009.12.008. PMID 20138767.
- ↑ Bénit, Paule; Goncalves, Sergio; Dassa, Emmanuel Philippe; Brière, Jean-Jacques; Rustin, Pierre (2008). "The Variability of the Harlequin Mouse Phenotype Resembles that of Human Mitochondrial-Complex I-Deficiency Syndromes". PLoS ONE. 3 (9): e3208. Bibcode:2008PLoSO...3.3208B. doi:10.1371/journal.pone.0003208. PMC 2527683. PMID 18791645.
- ↑ Joza, Nicholas; Susin, Santos A.; Daugas, Eric; Stanford, William L.; Cho, Sarah K.; Li, Carol Y. J.; Sasaki, Takehiko; Elia, Andrew J.; Cheng, H.-Y. Mary; Ravagnan, Luigi; Ferri, Karine F.; Zamzami, Naoufal; Wakeham, Andrew; Hakem, Razqallah; Yoshida, Hiroki; Kong, Young-Yun; Mak, Tak W.; Zúñiga-Pflücker, Juan Carlos; Kroemer, Guido; Penninger, Josef M. (2001). "Essential role of the mitochondrial apoptosis-inducing factor in programmed cell death". Nature. 410 (6828): 549–54. doi:10.1038/35069004. PMID 11279485.